They’re everywhere in longevity circles. Some of the science is genuinely exciting. But the gap between what’s promised and what’s proven is wider than most people realise.

Why Everyone Is Talking About Peptides
If you’ve spent any time in health and fitness spaces over the past few years, you’ve probably heard the word “peptides” thrown around with increasing confidence. Podcasts feature them. Biohacking communities swear by them. Your mate at the gym may have casually mentioned he’s “on BPC” for a dodgy shoulder.
The enthusiasm is understandable. The idea that a small, targeted molecule could accelerate healing, improve sleep, boost growth hormone, or slow ageing is deeply appealing — especially when the anecdotal reports sound so convincing.
But as a GP working in preventive and longevity medicine, I sit in an uncomfortable middle ground on this topic. I’m genuinely interested in the science. Some of it is promising. I also can’t ignore the fact that the gap between the preclinical research and what’s being sold to patients is, in many cases, enormous. And that gap matters when you’re injecting something into your body.
So this is my attempt to give you a clear-eyed, honest overview: what peptides actually are, which ones are generating the most interest, where the evidence stands, and how I think about them in clinical practice.
What Actually Are Peptides?
At the most basic level, a peptide is just a short chain of amino acids — the same building blocks that make up proteins. The distinction is size: proteins are long, complex chains (think hundreds or thousands of amino acids folded into intricate shapes), while peptides are shorter, typically between 2 and 50 amino acids in length.
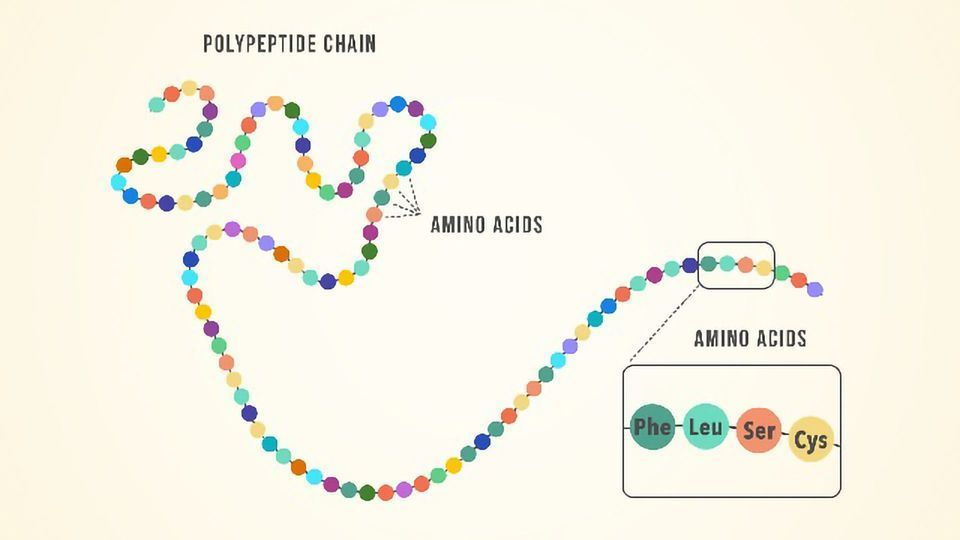
Your body produces thousands of peptides naturally. Insulin is a peptide. So is oxytocin. So are the endorphins that give you a runner’s high. These molecules act as signalling agents — they bind to specific receptors on cells and trigger particular biological responses. They’re essentially chemical text messages, telling cells what to do.
The peptides generating buzz in the longevity and performance world are synthetic versions of these signalling molecules, or novel sequences designed to mimic or amplify specific natural processes. The appeal is precision: rather than a broad-acting drug that affects multiple systems, a peptide theoretically targets a specific receptor and produces a specific effect.
That’s the theory, anyway. The reality is more complicated.
The Ones You’ll Hear About Most
There are dozens of peptides circulating in the wellness space, but a handful dominate the conversation. Here are the marquee names and what the interest is about.
BPC-157 (Body Protection Compound-157). This is probably the most talked-about peptide in the healing and recovery space. It’s a synthetic fragment of a protein found in human gastric juice, and in animal studies it has shown remarkable effects on tissue repair — tendon healing, gut lining restoration, reduction of inflammation, and even neuroprotective properties. The anecdotal reports from people using it for injuries are often glowing. The caveat: virtually all the evidence comes from rodent studies. There are no published randomised controlled trials in humans. We’re extrapolating from rats to people, and that’s a leap that doesn’t always land.
CJC-1295 / Ipamorelin. These are typically used in combination as growth hormone secretagogues — meaning they stimulate your pituitary gland to release more of your own growth hormone, rather than injecting synthetic growth hormone directly. The appeal is a more physiological, pulsatile pattern of GH release with fewer side effects than exogenous GH. There’s reasonable mechanistic plausibility here, and some clinical data exists for CJC-1295 specifically, but long-term safety data in healthy people using them for anti-ageing purposes is essentially absent.
Thymosin Beta-4 (Tβ4). Another healing and tissue-repair peptide, with research interest in wound healing, cardiac tissue repair, and anti-inflammatory effects. Again, animal data is intriguing. Human evidence is limited. It gained some notoriety when it surfaced in sports doping investigations, which tells you something about the perceived performance benefit — but perception and proof are different things.
GLP-1 receptor agonists (semaglutide, tirzepatide). Technically peptide-based medications, though they’ve entered a different category entirely because they’ve been through the full regulatory process. These are the drugs behind the weight loss revolution of the past few years, and they’re worth mentioning because they illustrate what happens when a peptide actually completes rigorous Phase III clinical trials: you get clear evidence of efficacy, a defined side effect profile, and regulatory approval. They’re the exception that highlights the rule. More about GLP-1 here.
Beyond these headline names, the broader peptide landscape includes categories like antimicrobial peptides, melanocortin-based peptides (such as PT-141 for sexual dysfunction and melanotan for tanning), and various mitochondrial-targeted sequences. The common thread is that most have interesting mechanistic rationale, limited or no human trial data, and a great deal of enthusiasm running ahead of evidence.
The Chinese EV Problem
I find the best way to explain my position on peptides to patients is with an analogy.

Over the past few years, a wave of Chinese-manufactured EV has entered the Australian market. Some of them are genuinely impressive. They’re packed with features — huge touchscreens, advanced driver-assist technology, premium finishes — often at a price point that undercuts the established brands by 30 to 40%. On paper and in a showroom, they look like an absolute bargain.
But when you go to check the ANCAP safety rating, there’s a gap. Some of these vehicles haven’t been through the independent crash testing that Australians rely on to know whether a car will protect their family in an accident. That doesn’t necessarily mean the car is unsafe. It might perform beautifully in a collision. But you don’t know. The testing hasn’t been done. And the testing is the part that matters, because it’s the difference between a manufacturer’s claim and an independently verified result.

Peptides are in a very similar position. Many of them come with impressive-looking features: fascinating mechanisms of action, compelling animal data, and a flood of enthusiastic testimonials. But for most of them, the human equivalent of an ANCAP safety rating — rigorous, independent, controlled clinical trials — simply doesn’t exist yet.
That doesn’t mean they’re dangerous. It doesn’t mean they don’t work. It means we don’t have the standard of evidence that I’d normally require before recommending something to a patient. And “it probably works and is probably safe” is a different sentence from “we’ve tested this in thousands of people and here’s what we found.”
The Pitfalls People Don’t Talk About
Beyond the evidence gap, there are several practical concerns that rarely make it into the podcast conversations.
Quality and purity. Most peptides used outside of clinical trials are sourced from compounding pharmacies or, increasingly, from overseas suppliers of variable quality. Unlike registered pharmaceuticals, there is no standardised manufacturing oversight for many of these products. Independent analyses have found peptide products containing less active ingredient than labelled, different peptides than labelled, or bacterial contamination. You might be injecting exactly what you think you’re injecting. You might not be.

Dose extrapolation. The doses used in most peptide protocols are extrapolated from animal studies using body-surface-area conversions or borrowed from small pilot studies. This is educated guesswork, not precision dosing. The therapeutic window — the range between an effective dose and a harmful one — is often unknown.
Unknown long-term effects. Growth hormone secretagogues, for example, raise a question that deserves honest acknowledgement: chronically elevated growth hormone and IGF-1 levels are associated in observational data with increased cancer risk. Does pulsatile stimulation via peptides carry the same risk as sustained elevation? Probably not to the same degree, but we genuinely don’t know. The long-term safety studies haven’t been done.
Regulatory grey zones. The regulatory landscape for peptides in Australia and New Zealand is evolving and, frankly, confusing. Some peptides are available through compounding pharmacies under prescriber authority. Others exist in a legal grey area. The rules change periodically, and what’s accessible today may not be tomorrow. This uncertainty isn’t just an inconvenience — it reflects the fact that regulators themselves aren’t yet confident about the risk–benefit profile of these compounds.
Where I Stand
I think peptides are one of the more interesting frontiers in medicine right now. The underlying science — the idea that we can use targeted signalling molecules to influence specific biological processes — is sound, and some of the preclinical data is genuinely exciting.
But I won’t prescribe what I can’t verify.
That’s not a philosophical position. It’s a practical one. When I prescribe testosterone, or a statin, or a GLP-1 receptor agonist, I can point to large-scale human trials that tell me what to expect: the likely benefit, the known side effects, the populations it works best in, and the monitoring required. That evidence base is what allows me to have an honest, informed conversation with a patient about whether a treatment is right for them.
I find it ironic how the worlds of Covid-19 vaccine hesitancy and the peptide craze sit on opposite ends of the scientific caution spectrum yet share a curious overlap in human behavior. On one hand, millions resisted a rigorously tested, population-wide intervention proven to save lives, often citing fear of long-term unknowns or mistrust of “big pharma.” On the other, the same demographic eagerly chases peptides—novel, largely experimental molecules touted to optimize longevity, fitness, or performance—with far less evidence and minimal regulatory oversight.
The paradox lies in the selective valuation of risk: well-studied vaccines provoke skepticism, while fringe biohacks inspire devotion. It’s a testament to how desire, social signaling, and the allure of cutting-edge science often outweigh rational appraisal of safety and efficacy.

For most peptides, I can’t do that yet. What I can do is stay across the emerging evidence, maintain an open mind, and be ready to incorporate new tools as the data matures. I’d rather be twelve months behind the hype curve and confident in what I’m recommending than twelve months ahead of it and guessing.
If you’re considering peptides, here’s my advice. Be curious, but be critical. Ask your provider what level of evidence supports the peptide they’re recommending, and whether that evidence comes from human trials or animal models. Ask where the product is sourced and whether it’s been independently tested. Be wary of anyone who speaks about peptides with absolute certainty — the honest answer on most of them is “promising but unproven.”
And above all, don’t let peptides distract from the interventions we know work. The unsexy foundations — sleep, exercise, nutrition, metabolic health, hormonal optimisation with proven therapies — will always deliver more reliable results than the most exciting unproven molecule.
The Chinese car might turn out to be brilliant. But I’d still want to see the crash test results before I put my family in it. I'll go with the Toyota.




