A Bloke's Guide to GLP-1 Agonists. They're not just "weight loss jabs." Here's what these drugs are actually doing inside your body — and why that matters more than the number on the scale.
If you've heard of Ozempic, you've probably heard someone call it "the skinny jab." That framing has done these medications a massive disservice. It's like calling statins "cholesterol pills" and leaving out the part where they reduce your risk of dying from a heart attack.
GLP-1 receptor agonists — semaglutide (Ozempic, Wegovy), liraglutide (Saxenda), and tirzepatide (Mounjaro) — are among the most significant pharmacological developments in metabolic medicine in decades. And the pipeline behind them is about to make the current generation look like a warm-up act.
Here's what you actually need to know.
The Two-Minute Version
GLP-1 medications work by amplifying your body's natural appetite and glucose-regulating signals. Their benefits extend well beyond weight loss into cardiovascular protection, metabolic health, and liver function. They're safe when prescribed and monitored by a doctor, and the side effects are manageable with proper titration and nutritional support.
Oral versions are imminent. Retatrutide — the triple agonist — is showing results that rival bariatric surgery and could be approved by 2027. But until it is, sourcing it from grey-market suppliers is a genuinely dangerous bet on an unknown substance with an unknown safety profile.
Talk to your GP. Get the prescription. Do it properly.
What GLP-1 Medications Do Inside Your Body
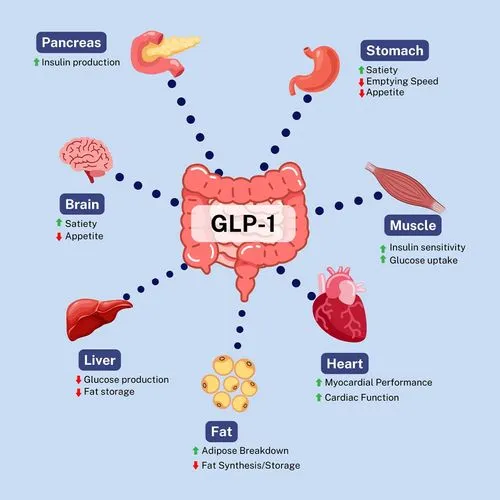
GLP-1 (glucagon-like peptide-1) is a hormone your gut naturally releases after eating. It does three main things: tells your pancreas to release insulin, slows down how quickly food leaves your stomach, and sends signals to your brain that you're full.
The problem is that natural GLP-1 gets broken down within minutes. GLP-1 receptor agonists are synthetic versions engineered to last much longer — days, in the case of semaglutide — so the effects are sustained.
When you take a weekly semaglutide injection, you're essentially amplifying a signal your body already produces. Your pancreas responds more efficiently to glucose (without the risk of hypoglycaemia if you don't have diabetes). Your stomach empties more slowly, so you feel full sooner and stay full longer. And crucially, the drug crosses the blood-brain barrier and acts on appetite centres in the hypothalamus, fundamentally reducing your drive to eat. This isn't willpower in a syringe — it's a correction of the neurohormonal signals that drive overconsumption.

Tirzepatide (Mounjaro) goes a step further. It's a dual agonist, targeting both GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors. The addition of GIP agonism appears to amplify the metabolic effects, which is why tirzepatide has consistently produced greater weight loss than semaglutide in head-to-head comparisons.
Benefits Beyond Weight Loss
This is where most blokes stop reading, and it's exactly where they should start paying attention.
GLP-1 medications have demonstrated reductions in major adverse cardiovascular events in people with established heart disease and type 2 diabetes. That's not a theoretical benefit — it's hard endpoint data from trials involving tens of thousands of people. We're talking about fewer heart attacks, fewer strokes, fewer cardiovascular deaths.
Beyond the ticker, the evidence is stacking up across multiple systems. Improved blood glucose control and insulin sensitivity, even in people who don't meet the diagnostic threshold for diabetes. Reductions in liver fat — significant for the growing number of blokes with metabolic dysfunction-associated steatotic liver disease (what used to be called fatty liver). Improvements in blood pressure, triglycerides, and inflammatory markers. Emerging evidence of benefits for sleep apnoea and chronic kidney disease.
For a bloke in his 40s or 50s carrying 15–20 extra kilos around the middle with a blood pressure that's "borderline" and an HbA1c that's "on the high side of normal," a GLP-1 agonist isn't just trimming the waistline. It's addressing the entire metabolic cluster that drives the leading causes of death in Australian men.
Dosing and Administration
All GLP-1 agonists follow a titration schedule — you start low and increase the dose gradually over weeks to months. This isn't optional. The slow ramp-up is specifically designed to let your gut adapt and minimise the side effects that cause most people to quit.
For semaglutide (Ozempic/Wegovy), you typically start at 0.25mg weekly for four weeks, then increase to 0.5mg, then 1.0mg, potentially up to 2.4mg for weight management. Each step is held for at least four weeks before moving up. The injection is subcutaneous — a tiny needle into the belly, thigh, or upper arm. Most people describe it as barely noticeable.
Tirzepatide follows a similar pattern: starting at 2.5mg weekly and titrating up through 5mg, 7.5mg, 10mg, 12.5mg, to a maximum of 15mg.
The temptation to skip ahead or start at higher doses is real — and it's a mistake. The GI side effects that make the news (nausea, vomiting, the infamous "Ozempic face") are almost always a function of going too hard too fast or not supporting the treatment with adequate nutrition and protein intake.
Side Effects: The Honest Version
The most common side effects are gastrointestinal — nausea, vomiting, diarrhoea, constipation, and abdominal discomfort. For most people, these are worst during the titration phase and settle as the body adjusts. Eating smaller meals, staying hydrated, and avoiding fatty or greasy foods helps significantly.
Less common but worth knowing about: gallstones (the rapid weight loss can trigger gallstone formation, just as it does after bariatric surgery), pancreatitis (rare but serious — persistent severe abdominal pain needs medical attention), and injection site reactions. There's a theoretical concern about thyroid C-cell tumours based on animal studies, which is why these medications are contraindicated in anyone with a personal or family history of medullary thyroid carcinoma.
Muscle loss. This question deserves a straight answer. Yes, when you lose significant weight with any method — medication, surgery, or diet — some proportion of that loss will be lean mass. The answer isn't to avoid the medication; it's to pair it with resistance training and adequate protein intake (1.6–2.2g per kilogram of body weight daily). This is non-negotiable for any bloke on a GLP-1 agonist.
Mental health and suicidality. In late 2025, the TGA updated warnings across all GLP-1 medications to flag a potential risk of suicidal thoughts or behaviours, advising doctors to monitor for mood changes. The evidence isn't sufficient to prove a causal link, and the numbers are small relative to millions of prescriptions, but the TGA noted a complex interplay between mental illness, metabolic conditions, and the psychological impact of rapid weight loss. If you have a history of depression or anxiety, make sure your GP knows before you start — and flag any mood changes early.
Fertility and "Ozempic babies." Weight loss can significantly boost fertility in both men and women — each BMI point above 29 reduces female fertility by roughly 5%, and obesity suppresses male hormones responsible for sperm production. GLP-1 medications may also reduce the absorption of oral contraceptive pills by slowing gastric emptying, particularly when starting or increasing the dose. The practical upshot: if you or your partner are on a GLP-1 agonist, use reliable contraception (preferably non-oral) unless pregnancy is the plan — and if it is the plan, these medications should be stopped at least two months before trying to conceive.
Are They Safe?
In a word: yes — with appropriate medical supervision. These medications have been through Phase 3 clinical trials involving tens of thousands of participants, they're approved by the TGA in Australia, and they're manufactured under strict pharmaceutical standards. The safety profile is well-characterised and the serious risks are known and manageable.
That safety assurance, however, only applies to TGA-approved products prescribed by a doctor and dispensed by a pharmacy.
In Australia, GLP-1 agonists are PBS-subsidised for type 2 diabetes (around $31.60 per script for general patients). For weight management without a diabetes diagnosis, they're typically a private prescription — anywhere from $130 to $400+ per month depending on the drug and dose. That cost barrier is real, and it's pushing some people toward alternatives that carry genuine risk. More on that shortly.
What's Coming Next
Oral GLP-1 Agonists
The injection barrier is about to disappear. Lilly is awaiting an FDA decision on orforglipron, an oral GLP-1 receptor agonist that has succeeded in Phase 3 trials for both obesity and type 2 diabetes. High-dose oral semaglutide formulations are also in development. Within a year or two, the weekly jab may become a daily tablet for many patients — which will fundamentally change the accessibility conversation.
Retatrutide: The Triple Agonist
This is the one generating the most excitement in metabolic medicine. Retatrutide is a triple hormone receptor agonist that mimics three gut hormones — GLP-1, GIP, and glucagon — and in its first Phase 3 trial, participants on the highest dose lost an average of 28.7% of their body weight over 68 weeks. To put that in context, that's approaching the results typically seen with bariatric surgery.
In the Phase 2 trial, at the highest dose, more than 9 in 10 participants lost at least 10% of their body weight, nearly two-thirds lost 20% or more, and a quarter lost 30% or more. The addition of glucagon receptor agonism appears to increase energy expenditure on top of the appetite suppression — your body burns more, not just eats less.
Seven additional Phase 3 trials are expected to complete in 2026 , covering obesity, type 2 diabetes, sleep apnoea, chronic low back pain, liver disease, and cardiovascular outcomes. Analysts predict a potential approval in 2027.
A Word on the Unapproved Peptides
Here's where we need to be blunt. Retatrutide is not approved anywhere in the world. It is still an investigational drug in clinical trials. Everyone taking it outside of those trials is essentially taking part in an unregulated experiment. We also cover the conversation around peptides in another article
Despite that, it's already available on grey-market peptide sites, usually labelled "for research purposes only" — a legal fig leaf that fools nobody. Independent testing has found that at least 20% of unapproved peptides sold online are mislabelled — vials claiming to contain retatrutide that actually contain semaglutide, or different compounds entirely. The FDA has issued warning letters to companies illegally selling products containing retatrutide, and has stated that retatrutide cannot legally be used in compounding.
The risks here aren't theoretical. You don't know what's in the vial. You don't know the dose. There's no sterile manufacturing guarantee. And there's no medical supervision if something goes wrong. The Phase 3 data also flagged a new safety signal — dysesthesia (abnormal sensory processing where normal touch is perceived as painful) — in up to 21% of patients on the highest dose, a side effect that wasn't seen in the earlier Phase 2 trial. That's exactly the kind of finding that emerges through proper clinical research — and exactly what you miss when you're injecting powder from an unlabelled vial you bought through a TikTok link.
If you wouldn't buy your blood pressure medication from an Instagram ad, don't buy your metabolic medications from a peptide vendor. The approved alternatives — semaglutide, tirzepatide — are available now, through your GP, with proper medical oversight and a known safety profile. Retatrutide will almost certainly be worth the wait.



