Understanding Alzheimer's: the 15-year head start you didn't know you had
Ten years ago, if your old man started forgetting things, the medical conversation was basically: "Well, it's either old age, or it's Alzheimer's, and we can't really tell the difference until he gets worse. And even then there's not much we can do."
That conversation has quietly changed. The shift isn't primarily about new drugs (although they exist, imperfectly). It's about the fact that Alzheimer's is now something we can see coming — years, sometimes decades, before the first symptom shows up. And that changes what "early intervention" actually means.
If you're somewhere between 35 and 55, you're right in the window where this matters. Not because you're about to get diagnosed. Because the biology of Alzheimer's is laying down its foundation right now, and for the first time, we've got tools to do something about it.
What Alzheimer's actually is
Quick terminology: dementia is the syndrome, Alzheimer's is one cause (there are over a hundred others). Alzheimer's accounts for around 70% of dementia cases. Worth keeping straight.
The biology has three main characters.
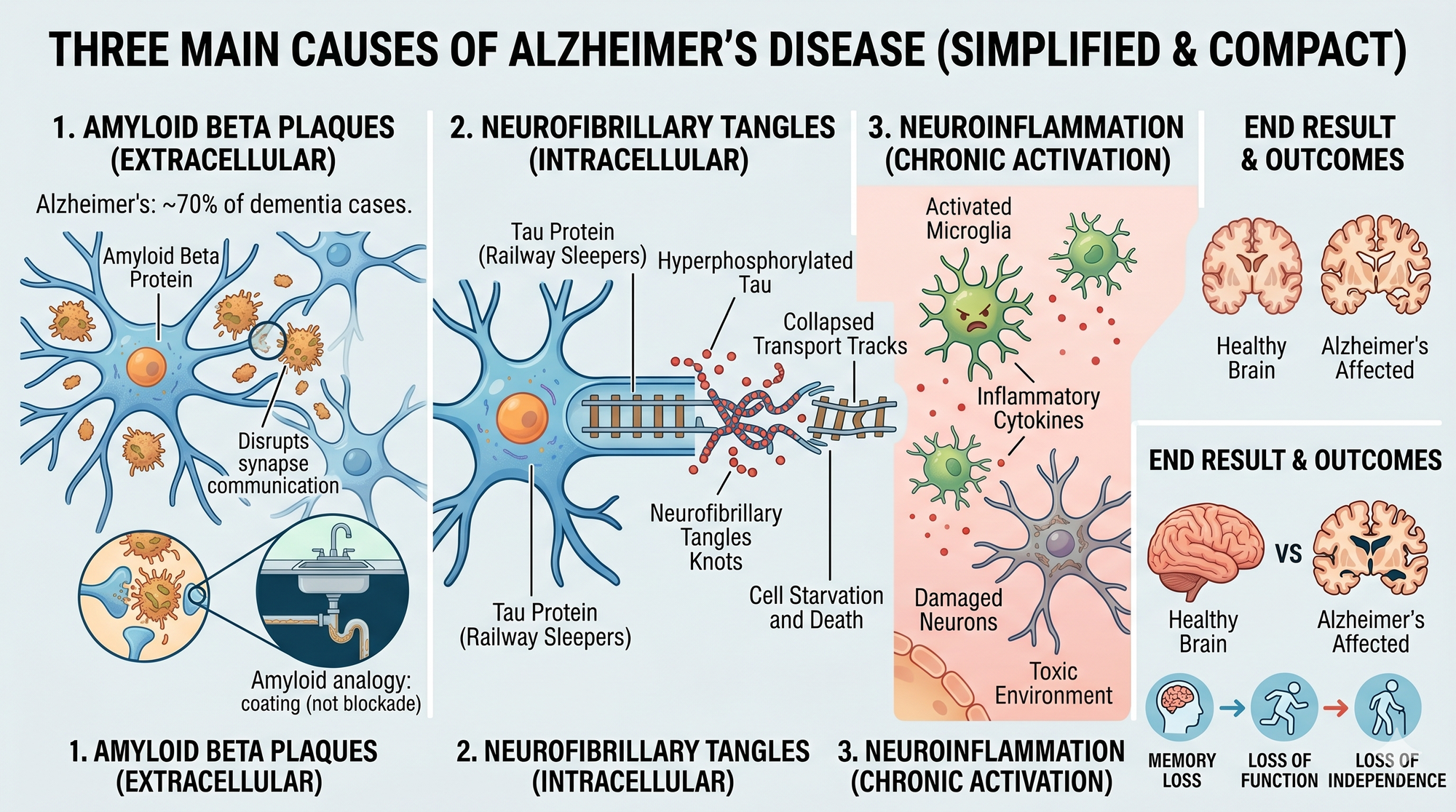
Amyloid beta plaques are sticky protein fragments that clump together in the spaces between brain cells. Imagine the plumbing under your kitchen sink — fat and bits of food slowly coating the inside of the pipe. Amyloid does something similar in brain tissue: less about physical blockage, more about disrupting cell-to-cell communication.
Neurofibrillary tangles are made of a protein called tau. Tau normally acts like railway sleepers — it stabilises the transport tracks inside each neuron so nutrients and signals can move along. When tau gets hyperphosphorylated (too many phosphate groups stuck on), it twists into knots and those transport lines collapse. The cell starves from the inside out.
Neuroinflammation is the background hum. Microglia — the brain's immune cells — get chronically activated and throw off inflammatory cytokines that damage neighbouring neurons.
End result: synapses die, neurons shrink, whole brain regions atrophy. The folds thin, the valleys widen. Memory goes first. Then function. Then independence.
The paradigm shift: the 15-year runway
Here's the insight that changed everything. By the time someone has noticeable memory problems, the disease has been running for 15 to 20 years.
The Alzheimer's Association now stages AD on a continuum from 0 to 6:
- Stage 0 — Deterministic genes present, biomarkers still in the normal range, no symptoms
- Stage 1 — Preclinical AD. Biomarkers abnormal (amyloid or tau detectable), daily life completely unaffected
- Stage 2 — Mild detectable cognitive change, minimal impact on daily function. Where people start noticing something's off
- Stage 3 — Cognitive impairment with early functional impact. Mild cognitive impairment (MCI) territory
- Stages 4 to 6 — Mild, moderate, and severe dementia
Amyloid changes first. Tau follows. Brain structure starts shifting on MRI. Then memory. Then function. On the research curves, the biomarkers go abnormal years before clinical function starts to drop.
In plain English: Alzheimer's isn't an event, it's a decades-long process. And the part where you can actually change the trajectory sits mostly in your 40s and 50s — long before anyone in the family has started worrying about Dad.
Subjective cognitive decline: the symptom people dismiss
There's an entity in the neurology literature called subjective cognitive decline (SCD). It's what happens when someone notices their thinking isn't quite right — they can't find words the way they used to, they walk into a room and forget what they came for, they've started writing everything down because they can't trust the memory — but formal cognitive testing still comes back normal.
SCD used to get brushed off as "oh, it's just getting older." Sometimes it is. But it's increasingly recognised as a genuine early signal. People with SCD have roughly double the risk of progressing to MCI or dementia compared with people without it. It's noisy — not everyone with SCD progresses, and plenty of people without SCD do — but it matters.
If your dad in his 60s or 70s is quietly telling you his memory's not what it was, don't reflexively reassure him. That's a conversation for his GP. For you, in your 40s or 50s, same principle: if you have the feeling something's slipping, that's worth raising, not shrugging off.
What testing actually exists right now (AU and NZ)
This is where most of the change has happened in the last 18 months.
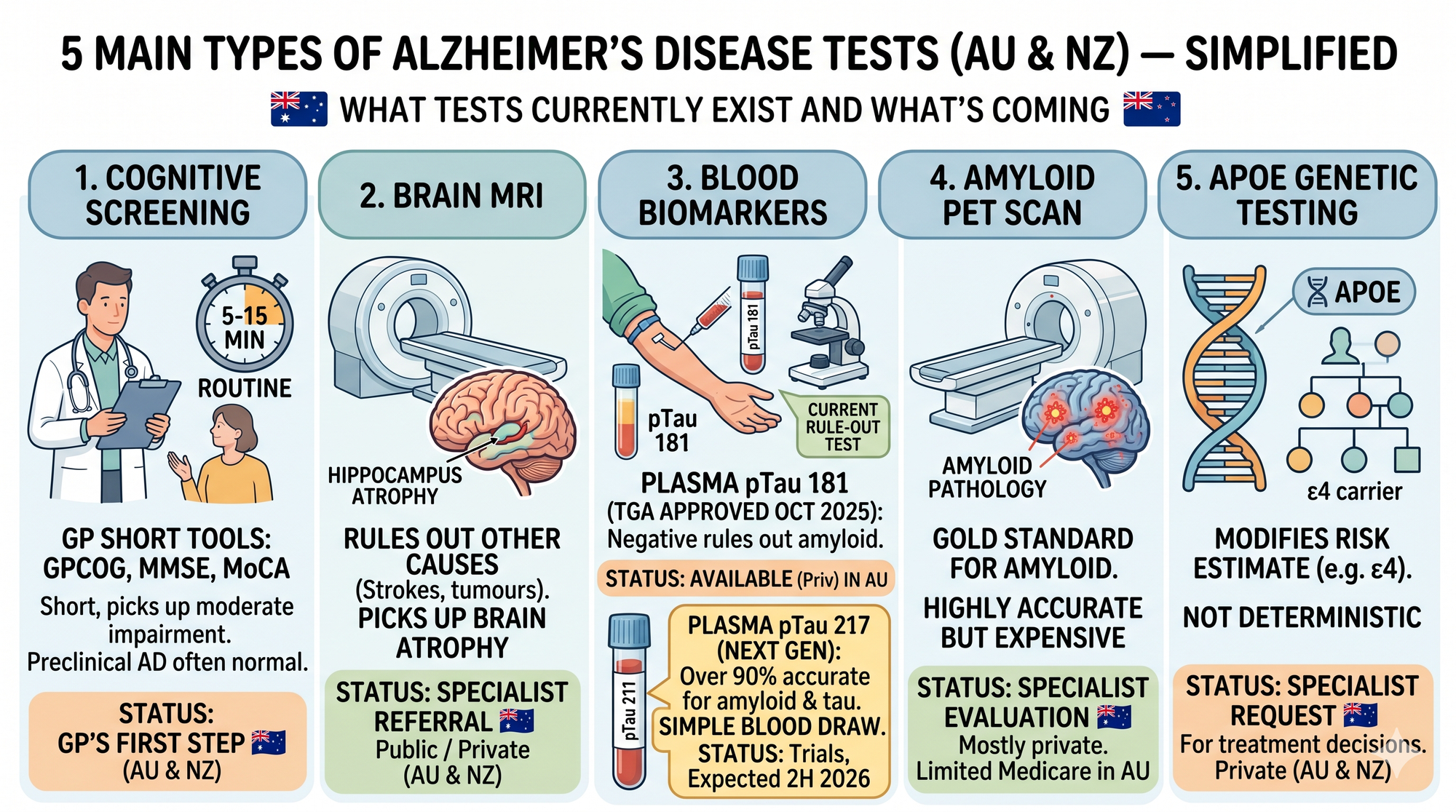
Cognitive screening. Your GP has several short tools — the GPCOG (developed in Sydney), the Mini-Mental State Examination (MMSE), and the Montreal Cognitive Assessment (MoCA). These take 5 to 15 minutes, pick up moderate impairment reasonably well, and are fine for a first look. They're not great at catching the earliest changes — someone with preclinical AD will usually score normal — but they're the first step in any workup.
Brain MRI. Picks up hippocampal atrophy, white matter changes, and rules out other causes (strokes, tumours, normal pressure hydrocephalus). Part of the routine specialist workup for cognitive concerns. Available on referral through the public system, faster privately.
Blood biomarkers — the big new arrival. Roche's Elecsys plasma pTau 181 test was TGA-approved and listed on the Australian Register of Therapeutic Goods in October 2025 — the first Alzheimer's blood test available in Australia. It's a rule-out test, intended for adults aged 55 to 80 with subjective cognitive decline or suspected MCI. A negative result strongly argues against amyloid pathology. A positive result means further workup.
The next-generation assay is the one to watch. A plasma pTau 217 test — considered the more accurate biomarker — is expected in Australia in the second half of 2026. Compared with PET scans and lumbar puncture, plasma pTau 217 is over 90% accurate at identifying significant amyloid or tau pathology in the brain. That's a genuine leap — useful AD biomarker information from a simple blood draw rather than a PET scan or a spinal tap.
Amyloid PET scan. Still the gold standard for confirming amyloid pathology, but expensive (several thousand dollars privately), not widely Medicare-rebated outside specific pathways, and generally reserved for specialist evaluation — particularly if someone's being considered for one of the new disease-modifying drugs.
APOE genetic testing. Available on specialist request. Knowing your ApoE status can modify your risk estimate, but it's not deterministic — plenty of ApoE4 carriers never develop AD, and plenty of non-carriers do. Most specialists only order this if a treatment decision depends on it.
In New Zealand, most of this sits through the public memory clinic pathway with variable regional availability, or privately. Plasma pTau tests aren't yet widely available through Pharmac-funded pathology.
Important caveat: none of this is population screening for an asymptomatic 45-year-old. The tests exist for people with cognitive concerns being worked up for possible AD. But the pathway is opening up fast — and knowing it exists is the first step in using it when the time comes, whether that's for yourself in 20 years or your parents next year.
The biggest lever: the 14 risk factors
While the biomarker story is genuinely exciting, the single most actionable piece of evidence for men in their 40s and 50s is still the prevention one.
In 2024, the Lancet Commission updated its analysis: up to 45% of dementia cases worldwide could be prevented or delayed by addressing 14 modifiable risk factors across the life course. Two were newly added: vision loss and high LDL cholesterol.
The list, in order of population impact:
- Hearing loss (7%)
- High LDL cholesterol (7%)
- Less education in early life (5%)
- Social isolation in late life (5%)
- Depression (3%)
- Air pollution (3%)
- Traumatic brain injury (3%)
- Physical inactivity (2%)
- Diabetes (2%)
- Smoking (2%)
- Hypertension (2%)
- Visual loss (2%)
- Obesity (1%)
- Excessive alcohol (1%)
Read that list again as a 45-year-old. Hearing, LDL, depression, TBI, inactivity, diabetes, smoking, hypertension, obesity, alcohol. That's the same list your GP is already nagging you about for your heart.
Your coronary arteries and the small vessels feeding your hippocampus share most of the same neighbourhood. Look after one, you're looking after the other. The cardiologists and the neurologists have been quietly fighting the same war for years — it just took an epidemiological commission to spell it out.
The specificity that's new in 2024: mid-life risk factors carry the biggest load. Meaning 35 to 55 isn't too early to be thinking about this. It's the window.
Where treatment sits today
Three tiers, and it's worth knowing where each one fits.
Non-drug approaches — exercise, cognitive engagement, hearing aids, treating sleep apnoea and depression, social connection. Don't change the underlying disease, but genuinely improve day-to-day function. For anyone diagnosed, these are the foundation. For anyone trying to prevent diagnosis, they're the whole game.
Symptomatic drugs — the workhorses for 20+ years. Donepezil, galantamine, rivastigmine (cholinesterase inhibitors) and memantine (NMDA receptor). All four PBS-funded in Australia and Pharmac-funded in NZ under specific criteria. Modest, temporary lift in cognition. They tweak the neurotransmitter dial; they don't touch the amyloid or tau.
Disease-modifying therapies (DMTs) — the new category. Monthly IV infusions of monoclonal antibodies that clear amyloid plaques. Donanemab (Kisunla) TGA-approved May 2025, lecanemab (Leqembi) September 2025. Both slow progression by around 30–35% over 18 months — roughly six weeks of delayed decline, which is why the PBAC declined PBS listing. Neither is Pharmac-funded.
Out-of-pocket: A$40,000–$80,000 a year, plus imaging and monitoring. Only works in very early disease. Real risks — brain swelling or small bleeds in roughly 1 in 200 carefully monitored patients. ApoE4 homozygotes excluded.
The DMTs matter as proof of concept: clearing amyloid can slow Alzheimer's. The next generation is in trials now, some aimed at preclinical AD where they might prevent symptoms emerging at all. But for a reader in their 40s, the drugs aren't today's story. Early detection and the 14 modifiable risk factors are where the leverage sits.
What to do with all of this
If you're somewhere between 35 and 55, the play is pretty clear.
For yourself: treat the modifiable risks now. Know your blood pressure and your LDL, and treat them properly — not "when you get around to it." Protect your hearing — if you've had ringing after gigs for twenty years, get it checked, and take the earplugs to the next one. Wear a helmet on the bike, the MTB, the paddle board. Keep moving. Don't smoke. Keep alcohol at or below the NHMRC's 10 standard drinks a week. Get your sleep assessed if you snore or wake exhausted. Stay connected to your mates — the bloke who quietly drifts out of the group chat in his 50s is making a brain-health decision without realising it.
For the generation above you: if a parent is telling you their memory isn't what it was, take it seriously and get them to a GP. The workup has genuinely changed — cognitive testing, MRI, the new plasma pTau 181 blood test (with pTau 217 coming in late 2026), memory clinic referral where indicated. A proper diagnosis early matters even setting aside the drug question. It opens up planning, support services, and the chance to treat modifiable contributors (depression, sleep apnoea, medication review, alcohol).
The paradigm shift in Alzheimer's is that we've stopped treating it as an event that happens to old people, and started treating it as a biology that starts in mid-life, builds for decades, and now — for the first time — has both preventive tools and detection tools we can actually use.
The 15-year runway is a gift. Most people spend it asleep.



